Blog
Expert guidance on environmental monitoring, regulatory compliance, and cold chain management
![Securing your Laboratory During Extended Absences [Practical Guide]](/images/blog/securing-laboratory-absences.png)
Securing your Laboratory During Extended Absences [Practical Guide]
A comprehensive guide to protecting your laboratory equipment and samples during holidays, weekends, and other extended staff absences with proper monitoring and preparation protocols.

FDA 21 CFR Part 11 Compliance Guide for Environmental Monitoring
A comprehensive guide to achieving FDA 21 CFR Part 11 compliance with your environmental monitoring system. Learn about electronic records, audit trails, and validation requirements.

Hidden Costs: Why Chart Recorders Are Bleeding Your Lab Dry
Discover the true cost of maintaining traditional chart recorders versus modern digital monitoring systems, and learn why ATEK offers significant long-term savings and improved compliance.

USP 797 Temperature Monitoring Requirements for Sterile Compounding
Understanding the temperature and environmental monitoring requirements for sterile compounding under USP 797. Essential guidance for hospital and compounding pharmacies.

Cold Chain Monitoring Best Practices for Pharmaceutical Distribution
Essential best practices for maintaining cold chain integrity in pharmaceutical distribution. Learn about temperature monitoring, excursion management, and regulatory compliance.

The Right Partner for Your Critical Process: ATEK
Discover why ATEK is the trusted partner for cold chain monitoring and environmental parameter management in life sciences, offering expertise, compliance, and cutting-edge technology.

The High Cost of Procurement Processes (6x More Expensive)
Discover why traditional procurement processes can cost up to six times more than direct monitoring solutions like ATEK, with a return on investment in just four months.

The Importance of Monitoring Liquid Nitrogen Level in Cryogenic Tanks
Understand why monitoring liquid nitrogen level is as important as temperature, through a simple and effective culinary metaphor.
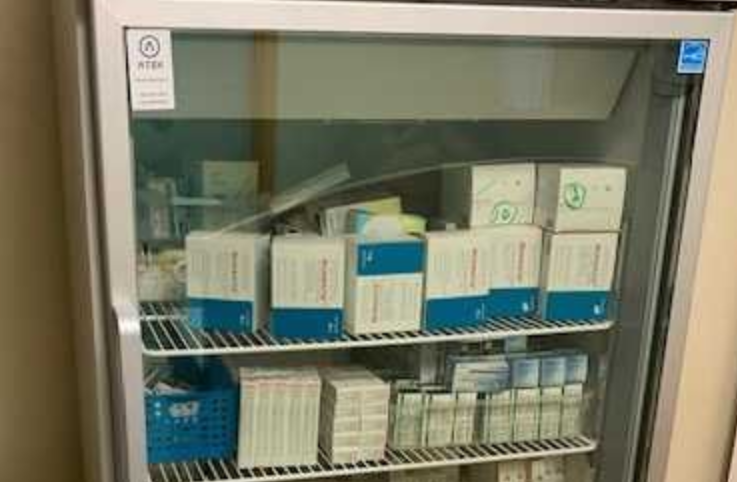
Product Spacing in Refrigerators
Discover why product spacing in vaccine refrigerators is crucial for maintaining uniform temperature and how to resolve temperature alert issues.

3 Advantages of Real-Time and Cloud
Discover the three main advantages of real-time monitoring and cloud data storage for GxP-regulated companies in the life sciences industry.

Guidelines for Vaccine Management Standards and Practices
Discover how the ATEK system facilitates compliance with Quebec's Guidelines for Vaccine Management Standards and Practices, point by point.

First Local Service Point Equipped with Negative Pressure Room
Discover how the CISSS of Montérégie-Centre integrated the first negative pressure room in a Local Service Point, with ATEK monitoring to fight infectious diseases.
Stay Ahead of Compliance
Get the latest insights on environmental monitoring and regulatory compliance delivered to your inbox.